 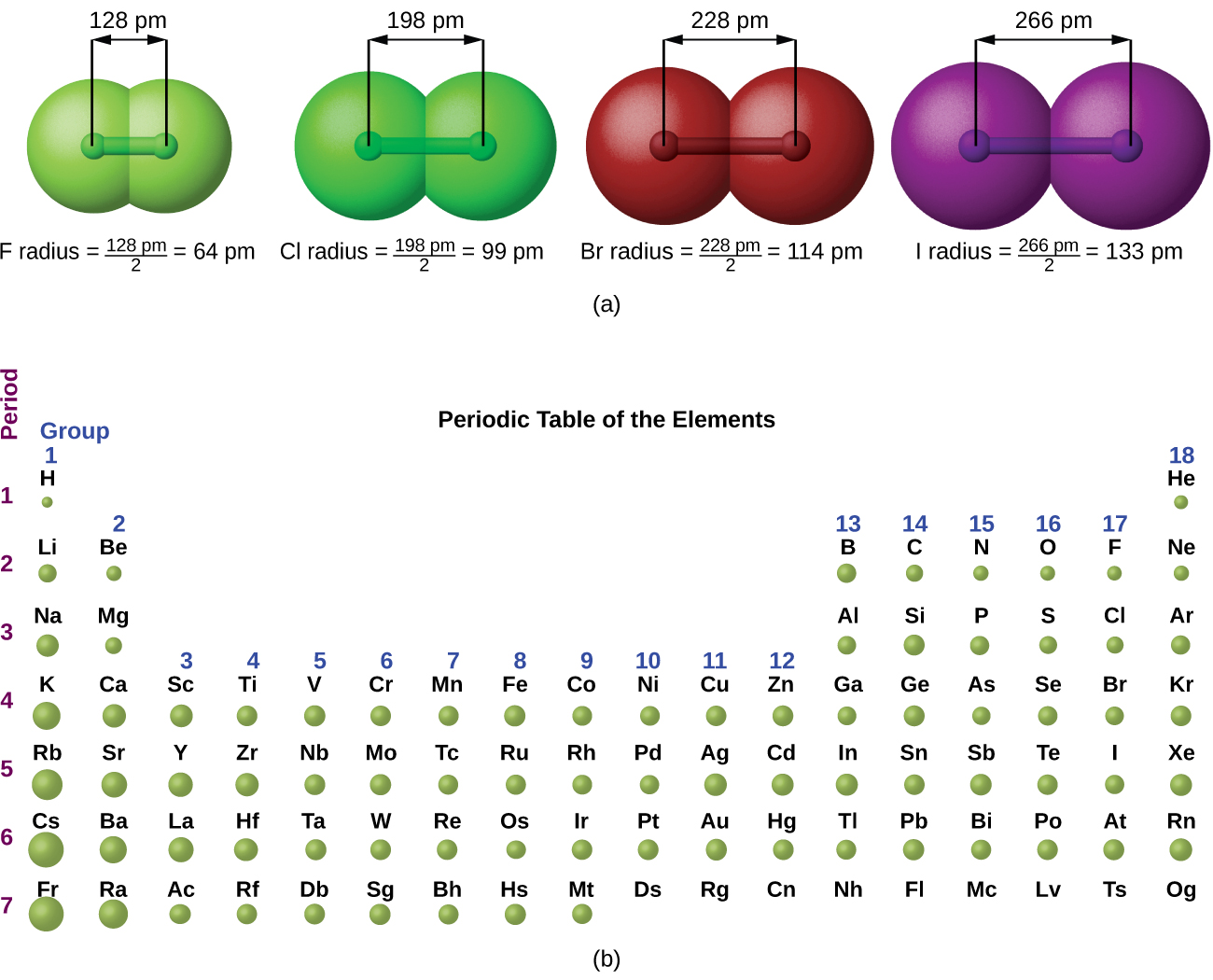
Electro Negativity of Bromine: 2.96 Atomic Volume of Bromine: 25.6 cm³/mol Name Origin of Bromine: Greek: brômos (stench). Three factors affect electronegativity: Atomic radius, nuclear charge and sheilding. The electron cloud forming the shell of an atom does not have any fixed shape which makes it difficult to determine the. Sulphur is closest to the most electronagative element, which is flourine. Measuring the atomic radii of chemical elements is a complicated task as the size of an atom is of the order of 1.2×10-10 m. From these data we predict the C - Br distance in CBr4 (or. Most characteristic isotope: Bromine-79 and bromine-81. An atomic radius is half the distance between adjacent atoms of the same element in a molecule. The element can be released from the bromides by treating them with a strong oxidizing agent such as chlorine. For example, the covalent radius of carbon is 0.77 and the covalent radius of bromine is 1.14. The salt silver bromide, AgBr, is used for making photographic emulsions and appears naturally in the mineral bromargyrite.īromine occurs in the form of compounds in small quantities in sea water and in natural salt deposits. Reaction with metals or metallic bases produces bromides such as sodium bromide, NaBr and potassium bromide, KBr which are used in medicine. In solution with water it forms a strong acid, hydrobromic acid. (A) Trends in the Atomic Radius of Elements in Period 2 number of occupied energy levels (electron shells) remains the same (2 occupied electron shells) across. 42) The number of valence electrons found in an atom of a Group A element is equal to. Hydrogen bromide, HBr, is a colorless gas. 41) The atomic radius of bromine is larger than the atomic radius of. 
It produces painful lesions when spilled on the skin. The one-half of the distance between the nuclei of identical atoms that are bonded together is called the atomic radius which increases down the group and decreases across the period.Bromine is an easily volatile, dark reddish-brown liquid with a strong disagreeable odor and an irritating effect on the eyes and throat. Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of bromine-80 (atomic. The horizontal rows in the periodic table are referred to as the period while the vertical columns are called the groups in the periodic table. The atomic radius varies with increasing atomic number, but usually increases because of increasing of. Therefore, the correct answer is option D, potassium is larger in atomic radius. The atomic radius of a chemical element is a measure of the size of its atom. group number 17 but atomic radius increases down the group in the periodic table hence chlorine is larger in atomic radius than bromine. While chlorine and bromine are present in the same group i.e. Similarly, potassium and bromine are present in the same period and as the atomic radius decreases from left to right in a periodic table, therefore, potassium is larger in atomic radius than bromine because potassium is located at the extreme left in the period while bromine is located at the extreme right in the period. Since potassium is located at the start of period 3, and bromine at the end of the same period, potassium will have a larger atomic radius than bromine, and thus the largest atomic radius of the four given atoms. Hence, potassium is larger in atomic radius than sodium. Likewise, bromine will have a larger atomic radius when compared with chlorine. Question: Question 12 0 / 1 point The atomic radius of bromine is smaller than the atomic radius of selenium because there are more energy levels in. group number one and atomic radius increases down the group in the periodic table. While sodium and potassium are present in the same group in the periodic table i.e. Also, atomic size decreases on going from left to right along the period in the periodic table, therefore, sodium is larger in atomic size than chlorine because sodium is present at extreme left in the period and chlorine is present at the extreme right in the period. Since sodium and chlorine are located in the same period i.e. 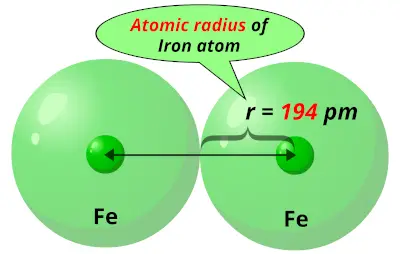 
Sodium, potassium belongs to group one of the periodic table and are called alkali earth metals, while chlorine and bromine belong to group seventeen of the periodic table and are called halogens. Hint: Due to the addition of electrons to the same shell in all atoms, the atomic radius decreases on moving from left to right across the period in a periodic table. Answer: > Bromine has what atomic radius than fluorine This sounds incomplete. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
